News
News
Press Release
Sysmex Launches an Assay Kit for Identifying p-Tau217 in the Blood, a Biomarker Related to Alzheimer's Disease
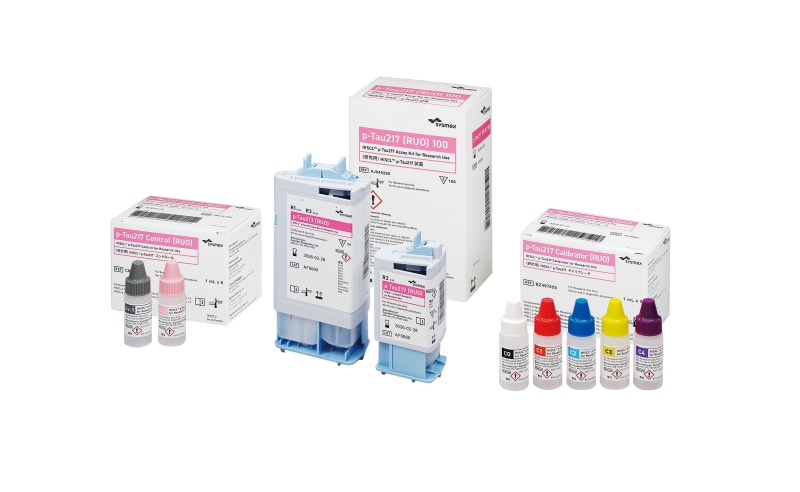
Sysmex Corporation (HQ: Kobe, Japan; President: Iwane Matsui) announced that it has launched “HISCL™ p-Tau217 Assay Kit for Research Use” (“the Product”), a reagent for research purposes to identify a biomarker1 related to Alzheimer's disease, namely blood p-Tau2172 levels, in Europe in April 2026. Following the launch in Europe, the Product will be rolled out sequentially in Japan, the United States, South Korea, and Taiwan, contributing to accelerated global data acquisition for Alzheimer’s disease research.
Going forward, Sysmex will support the creation of new knowledge through collaborative research, both domestically and internationally, and promote initiatives aimed at applying for and obtaining regulatory approval as an in vitro diagnostic product. This will lead Sysmex to establish less burdensome testing and diagnostic techniques for patients and to promote their early dissemination both domestically and internationally.
| Name of parameter | p-Tau217 kit | |
| Product name | HISCL™ p-Tau217 Assay Kit for Research Use | |
| Intended use | Quantitative measurement of p-Tau217 in plasma | |
| Manufacturer and seller | Sysmex Corporation | |
| Target market | Europe *To be released sequentially in Japan, the United States, South Korea, and Taiwan. |
|
| Launch | April 2026 | |
|
June 22, 2023 news release: “Sysmex Launches an Assay Kit to Identify Amyloid Beta (Aβ) Accumulation in the Brain, a Cause of Alzheimer’s Disease, Using a Small Amount of Blood”
https://www.sysmex.co.jp/en/news/2023/230622.html |
|||
|
August 29, 2023 news release: “Sysmex Begins Supplying Reagents for U.S. Based Laboratory Developed Test (LDT) Measuring Amyloid Beta (Aβ) in Blood” |
|||
| December 25, 2023 news release: “Sysmex Expands Sales of Its Reagents for Blood-based Amyloid β Testing to Europe” https://www.sysmex.co.jp/en/news/2023/231225.html |
|||
| 1 |
Biomarkers:
Internal indicators that provide clues about the state and changes in a disease, such as proteins found in the blood. |
||||||||
| 2 | p-Tau217: Abbreviation for phosphorylated tau-217. p-Tau217 refers to a molecular species in which the 217th site of the tau protein is phosphorylated, and which is being studied as an indicator related to the pathogenesis of Alzheimer's disease. |
||||||||
| 3 | Reagents for blood-based amyloid β testing: | ||||||||
|
|||||||||
| 4 | LDT: A laboratory developed test (LDT) is a type of in vitro diagnostic test that is designed, manufactured, and used within a single laboratory. |
||||||||
| 5 | HISCL™-Series: | ||||||||
|
|||||||||
About Sysmex Corporation
Sysmex Corporation, headquartered in Kobe, Japan, is a global leader in in vitro diagnostics. Since its foundation in 1968, Sysmex has focused on diagnostics as the core of its business, and today, it supports the health of people in over 190 countries and regions worldwide. Sysmex continues to innovate in diagnostics, and to collaboratively create unique values in the areas of personalized medicine and novel treatments, under its long-term vision of "Together for a better healthcare journey." Through its unique technology, solutions, and co-creation with various partners, Sysmex delivers new value and addresses the universal desire of people to live longer and healthier lives. For more information about Sysmex, please visit www.sysmex.co.jp/en/.
* "Healthcare journey" is a trademark of Sysmex Corporation, registered in Japan.
FOLLOW US ![]()
![]()
- The purpose of this news release is to communicate our business activities to our stakeholders. It may or may not include information about Sysmex's products or their research and development, but this is not intended for promotion, advertising or medical advice. The information contained in this news release is current as of the date of the announcement but may be subject to change without prior notice.
